Application of the SPR Biosensor in Drug Prototypes Discovery with Human Cytochrome P450(51)
as an Example
1Institute of Biomedical Chemistry, 10 Pogodinskaya str., Moscow, 119121 Russia; *e-mail: la-kaluzhskiy@yandex.ru
2Institute of Bioorganic Chemistry of the National Academy of Sciences of Belarus,
5/2 Acad. Kuprevich Street, Minsk, 220141 Belarus
3Elyakov Pacific Institute of Bioorganic Chemistry, Far Eastern Branch of Russian Academy of Sciences,
159, 100 Let Vladivostoka av., Vladivostok, 690022 Russia
Key words: surface plasmon resonance (SPR); screening; human cytochrome P450(51); drug prototypes
DOI:10.18097/BMCRM00055
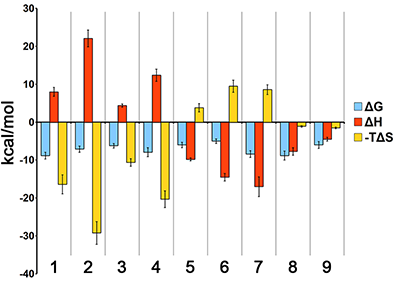
The development of the integral platform “From Gene to Lead”, consolidated computer methods, bioinformatics researches, and experimental approaches, significantly accelerated and optimized base structure search in the field of drug design. The necessity of the experimental verification of hundreds virtual structure hypothesis (results of molecular data base selections or de novo construction) requires demands the usage of the high-through out and sensitive methods for validation possible interaction between numerous of selected compounds and particular molecular targets and evaluation of affinity, kinetics and thermodynamics. Surface plasmon resonance (SPR) technology makes it possible to solve all these problems. In this article the methodical aspects of the optical SPR-biosensor usage in the field of drug prototypes selection are described using the human cytochrome P450(51) catalyzing one of the key step of cholesterol biosynthesis as an example.
|
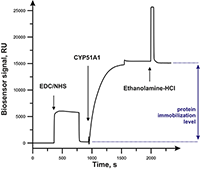
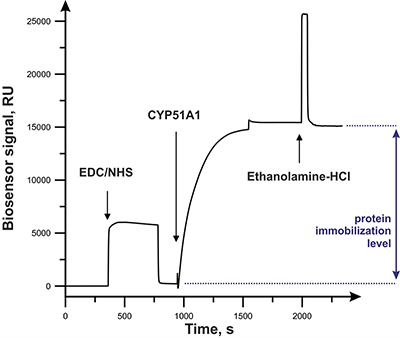
Figure 2.
Typical sensorgram of CYP51A1 immobilization. The beginning of solution injections is marked with arrows.
|
|
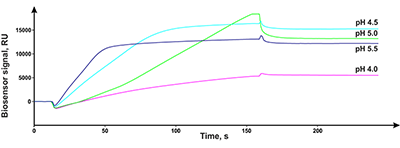
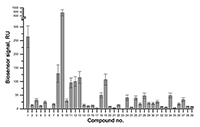
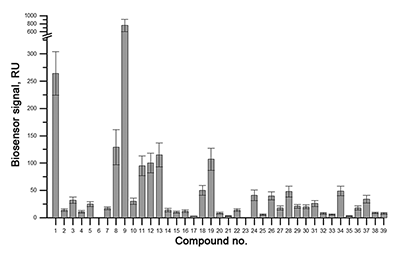
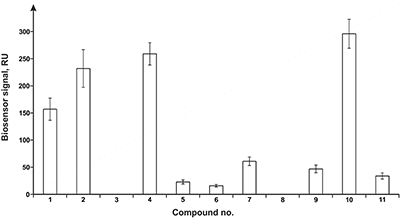
Figure 3.
Results of in vitro SPR screening of low-weight compound interactions
with CYP51A1 [4]. |
|
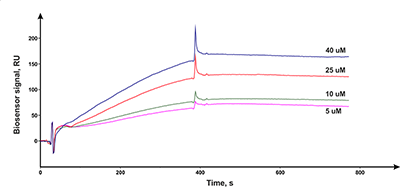
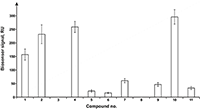
Figure 4.
Results of in vitro SPR screening of low-weight compound interactions
with CYP51A1 [5]. |
ACKNOWLEDGEMENTS
This study were performed within the framework of Program of Basic Scientific Research of Russian National Science Academies for 2013–2020 years (0518-2018- 0003) using the equipment of "Human Proteome" Core Facility of the Institute of Biomedical Chemistry which is supported by The Ministry of Education and Science of the Russian Federation under agreements №14.621.21.0017 (project ID RFMEFI62117X0017). Protein expression, spectrophotometric titration and enzyme assay were performed within the framework of the Belarusian State Program "High technologies and equipment" for 2016–2020 years (sub-program 1, “Innovative Biotechnologies – 2020”).
REFERENCES
- Ivanov, A.S., Veselovsky, A.V., Dubanov, A.V., Skvortsov, V.S. (2006). Bioinformatics platform development: from gene to lead compound. Methods in Molecular Biology, 316, 389-431. DOI
- Strushkevich, N., Usanov S.A., Park H.W. (2010) Structural basis of human CYP51 inhibition by antifungal azoles. Journal of Molecular Biology, 397(4), 1067-1078. DOI
- Gnedenko, O.V., Kaluzhskiy, L.A., Molnar, A.A., Yantsevich, A.V., Mukha, D.V., Gilep, A.A., Usanov, S.A., Stonik, V.A., Ivanov, A.S., Lisitsa, A.V., Archakov, A.I. (2013) The SPR-based biosensor test system for analysis of small compounds interaction with human cytochrome P450 51A1 (CYP51A1). Biochemistry (Moscow) Supplement. Series B, Biomedical Chemistry,7, 187–195. DOI
- Kaluzhskiy, L.A., Gnedenko, O.V., Gilep, A.A., Strushkevich, N.V., Shkel, T.V., Chernovetsky, M.A., Ivanov, A.S., Lisitsa, A.V., Usanov, A.S., Stonik, V.A., Archakov, A.I. (2014) Screening of human cytochrome P450(51) (CYP51A1) inhibitors: structural lanosterol analogues of plant and animal origin. Biochemistry (Moscow) Supplement. Series B, Biomedical Chemistry, 8, 349–360. DOI
- Kaluzhsky, L.A., Shkel, T.V., Ivanchina, N.V., Kicha, A.A., Grabovec, I.P., Gilep, A.A., Strushkevich, N.V., Chernovetsky, M.A., Medvedev, A.E., Usanov, S.A., Ivanov, A.S. (2017) Structural analogues of lanosterol from marine organisms of the class Asteroidea as potential inhibitors of human and Candida albicans lanosterol 14α-demethylases. Natural Product Communications, 12 (12), 1843-1846.
- Kicha, A.A., Kalinovsky, A.I., Levina, E.V., Stonik, V.A., Elyakov, G.B. (1983) Asterosaponin P1 from the starfish Patiria pectinifera. Tetrahedron Letters, 24, 3893–3896. DOI
- Kicha, A.A., Ivanchina, N.V., Gorshkova, I.A., Ponomarenko, L.P., Likhatskaya, G.N., Stonik, V.A. (2001) The distribution of free sterols, polyhydroxysteroids and steroid glycosides in various body components of the starfish Patiria (=Asterina) pectinifera. Comparative biochemistry and physiology. Part B, Biochemistry & Molecular Biology, 128, 43–52. DOI
- Kicha, A.A., Kalinovskii, A.I., Gorbach, N.V., Stonik, V.A. (1993) New polyhydroxysteroids from the Far Eastern sea star Henricia sp. Chemistry of Natural Compounds, 29, 206–210. DOI
- Kicha, A.A., Kalinovskii, A.I. (1993) Isolation of laeviuscoloside G from the starfish Henricia derjugini and correction of the structures of echinasterosides B1 and B2. Chemistry of Natural Compounds, 29, 547–548. DOI
- Kicha, A.A., Kapustina, I.I., Ivanchina, N.V., Kalinovsky, A.I., Dmitrenok, P.S., Stonik, V.A., Pal’yanova, N.V., Pankova, T.M., Starostina, M.V. (2008) Polyhydroxylated steroid compounds from the Far Eastern starfish Distolasterias nipon. Russian Journal of Bioorganic Chemistry, 34, 118–124. DOI
- Ivanchina, N.V., Kicha, A.A., Huong, T.T., Kalinovsky, A.I., Dmitrenok, P.S., Agafonova I.G., Long, P.Q., Stonik, V.A. (2010) Highly hydroxylated steroids of the starfish Archaster typicus from the Vietnamese waters. Steroids, 75, 897–904. DOI
- Myszka, D.G. (2000) Kinetic, equilibrium, and thermodynamic analysis of macromolecular interactions with BIACORE. Methods in Enzymology, 323, 325-340. DOI
- Ershov, P.V., Gnedenko, O.V., Molnar, A.A., Lisitsa, A.V., Ivanov, A.S., Archakov, A.I. (2012) Kinetic and thermodynamic analysis of dimerization inhibitors binding to HIV protease monomers by surface plasmon resonance. Biochemistry (Moscow) Supplement Series B: Biomedical Chemistry, 6(1), 94–97. DOI
- Trosken, E.R., Straube, E., Lutz, W.K., Volkel, W., Pattenm C. (2004) Quantitation of lanosterol and its major metabolite FF-MAS in an inhibition assay of CYP51 by azoles with atmospheric pressure photoionization based LC-MS/MS. Journal of the American Society for Mass Spectrometry, 15, 1216–1221. DOI
- Lepesheva GI, Waterman MR. (2007) Sterol 14α-demethylase cytochrome P450 (CYP51), a P450 in all biological kingdoms. Biochimica et Biophysica Acta General Subjects,1770, 467–477. DOI
- Lepesheva, G.I., Friggeri, L., Waterman, M.R. (2018) CYP51 as drug targets for fungi and protozoan parasites: past, present and future. Parasitology, 12, 1-17. DOI
- Gilli, P ., Gilli, G., Borea, P.A., Varani, K., Scatturin, A., Dalpiaz, A. (2005) Binding thermodynamics as a tool to investigate the mechanisms of drug-receptor interactions: thermodynamics of cytoplasmic steroid/nuclear receptors in comparison with membrane receptors. Journal of Medicinal Chemistry, 48(6), 2026-2035. DOI