The 40th Anniversary of the Institute of Physiologically Active Compounds of the Russian Academy of Sciences
Hydrogen Bond Contribution to Drug Bioavailability: cheminformatics approach
Institute of Physiologically Active Compounds of the Russian Academy of Sciences,
1 Severny proezd, Moscow region, Chernogolovka, 142432 Russia;*e-mail: raevsky@ipac.ac.ru
Key words: QSAR; HYBOT; hydrogen bond descriptors; bioavailability
DOI: 10.18097/BMCRM00060
A review, based mainly on own publications, is devoted to methods of investigation of “structure-bioavailability” relationships. The first part of this review contains information about classification of hydrogen bond descriptors, original 2D hydrogen bond thermodynamic descriptors, program HYBOT, original 3D hydrogen bonding potentials, original hydrogen bond surface area descriptors. The second part includes the results of applications of the above mentioned of hydrogen bond descriptors for prediction of bioavailability components such as lipophilicity, solubility in water and in physiological fluids, absorption and blood brain barrier permeability.
|
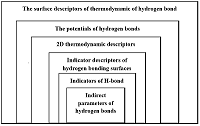
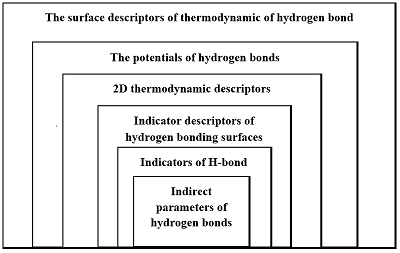
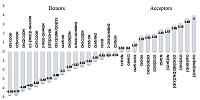
Figure 1.
Hierarchy of the information level of various hydrogen bond descriptors [10].
|
|
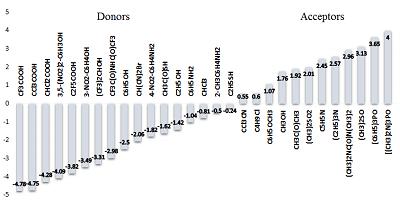
Figure 2.
TUnified scale of donor and acceptor factors of hydrogen bond (example for simple organic compounds).
|
|
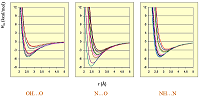
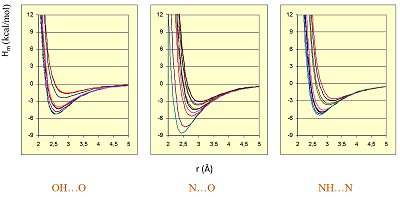
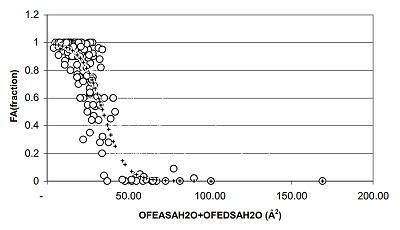
Figure 4.
The dependences of the fractions absorption (FA) from surface's HYBOT hydrogen bonds descriptors (OFEASA + OFEDSA).
|
|
CLOSE

|
Table 1. The simple and consensus AMP and LoReP models of solubility in water of 2,615 crystalline compounds.
|
|
CLOSE

|
Table 3.
Protocol of "CNS/not-CNS" classification of compounds by intuitive approaches and statistical methods. TP-correct recognition of "CNS", FN-incorrect recognition of "CNS", TN-correct recognition of "not-CNS", EP-misdiagnosis, "not CNS", SE-sensitivity (TP / TP + EN), SP- (TN / TN ++ EP), ACC- accuracy (TP + TN) / (TP + EN + TN + EP).
|
ACKNOWLEDGEMENTS
The work was performed within the framework of the state task for 2018 (the topic number 0090-2017-0020).
REFERENCES
- Pimentel, G. C., & McClellan, A. L. (1960). The hydrogen bond. Freeman: San Francisco, CA.
- Moore, T. S., & Winmill, T. F. (1912). CLXXVII.—The state of amines in aqueous solution. Journal of the Chemical Society, Transactions, 101, 1635-1676. DOI
- Latimer, W. M., & Rodebush, W. H. (1920). Polarity and ionization from the standpoint of the Lewis theory of valence. Journal of the American Chemical Society, 42(7), 1419-1433. DOI
- Varfolomeev, S. D., & Pozhitkov, A. E. (2000). Active centers of hydrolases: the main types of structures and the mechanism of catalysis. Vestnik Moskovskogo universiteta [Bulletin of Moscow University]. Part 2. Chemistry, 41(3), 147-156.
- Pauling, L., & Pauling, P. (1978). Chemistry [Russian translation]. Mir, Moscow, 299.
- Kubinyi H. (2001) Hydrogen bonding, the last mystery in drug design? In: Pharmacokinetic. Optimization in Drug Research (B.Testa , H.Van de Waterbeemd, G.Folkers R.Guy, eds.) Wiley - VCH, Weinheim and VHCA, Zurich, pp.513 - 524.
- Hamelink, J., Landrum, P. F., Bergman, H., & Benson, W. H. (1994). Bioavailability: physical, chemical, and biological interactions. CRC Press.
- U.S. Code of Federal Regulations. 21 CFR 320.1.
- Devillers, J., & Balaban, A. T. (Eds.). (2000). Topological indices and related descriptors in QSAR and QSPAR. CRC Press.
- Raevskij, O.A. (2015). Modelirovanie sootnoshenij “struktura-svojstva”, Dobrosvet, M. (in Russian)
- Raevskii, O. A., Avidon, V. V., & Novikov, V. P. (1982). Use of a unified scale of donor-acceptor interactions for the analysis of the similarity of the structures of biologically active compounds. Pharmaceutical Chemistry Journal, 16(8), 633-636. DOI
- Raevskii, O. A., & Novikov, V. P. (1982). Unification of the characteristics of donor-acceptor interactions in the framework of the problem of the structure-activity relationship. Khim.-Farm. Zh., 16(5), 583-586.
- Raevsky, O. A., Grigor'ev, V. Y., & Solov'ev, V. P. (1989). The Estimation of Donor-Acceptor Parameters in Biologically Active Compounds. Khim. Pharm. Zhurn.(Rus.), 23, 1294-1300.
- Martynov, I. V., & Raevsky, O. A. (1983). Physical-chemical approach to purposeful search of biological active substances. Vestnik akademii nauk SSSR, (7), 93-101.
- Martynov, I. V., & Raevskii, O. A. (1983). Estimation of electron-donor and acceptor ability of some active-centers in molecules of physiologically active compounds. Zhurnal vsesoyuznogo khimicheskogo obshchestva imeni di mendeleeva, 28(6), 716-717.
- Raevskii, O. A., Grigor'ev, V. Y., & Solov'ev, V. P. (1984). Evaluation of the electron-donor and electron-acceptor functions of ionized atoms and groups in biologically active substances on the basis of thermodynamic data. Pharmaceutical Chemistry Journal, 18(5), 327-331.
- Sapegin, A. M., Raevsky, O., Chistyakov, V. V., & Martynov, I. V. (1987). Donor-acceptor factor-analysis algorithm to depict molecules of biologically-active compounds. Khimiko-farmatsevticheskii zhurnal, 21(9), 1098-1102.
- Raevsky, O., & Sapegin, A. M. (1987). Developed physicochemical approach to recognition of physiologically active compound structures. Khimiko-farmatsevticheskii zhurnal, 21(11), 1338-1341.
- Sapegin, A. M., Razdolsky, A., Chistyakov, V. V., & Raevsky, O. (1987). structure recognition-realization of a physicochemical approach to the study of structure-activity-relationships. Khimiko-farmatsevticheskii zhurnal, 21(11), 1341-1344.
- Raevsky, O. A., Solotnov, A. F., & Solovyev, V. P. (1987). Electron-donating and electrophilic functions of физиологически physiological acting and model compounds. Journal of common chemistry, 57(6), 1241-1248.
- Raevskii, O. A., Grigoriev, V., Soloviev, V., & Martynov, I. V. (1988). Electron-acceptor enthalpy factors of phenols. Doklady Akademii Nauk SSSR, 298(5), 1166-1169.
- Raevsky, O. A., Grigor'ev, V. Y., Kireev, D. B., & Zefirov, N. S. (1992). Complete thermodynamic description of H‐bonding in the framework of multiplicative approach. Quantitative Structure‐Activity Relationships, 11(1), 49-63. DOI
- Raevsky, O. A. (1997). Quantification of non‐covalent interactions on the basis of the thermodynamic hydrogen bond parameters. Journal of physical organic chemistry, 10(5), 405-413. DOI
- Raevsky, O. A. (1997). Hydrogen bond strength estimation by means of the HYBOT program package. Computer‐Assisted Lead Finding and Optimization: Current Tools for Medicinal Chemistry, 367-378.
- Abraham, M. H., Ibrahim, A., Zissimos, A. M., Zhao, Y. H., Comer, J., & Reynolds, D. P. (2002). Application of hydrogen bonding calculations in property based drug design. Drug Discovery Today, 7(20), 1056-1063. DOI
- Ruelle, P. (1999). Towards a comprehensive non-ergodic treatment of H-bonds and hydrophobicity in real solutions: The mobile order and disorder theory. Perspectives in Drug Discovery and Design, 17(1), 61-96.
- Laurence, C., Brameld, K. A., Graton, J., Le Questel, J. Y., & Renault, E. (2009). The p K BHX database: toward a better understanding of hydrogen-bond basicity for medicinal chemists. Journal of medicinal chemistry, 52(14), 4073-4086. DOI
- Raevsky, O. A., Grigor’ev, V. J., & Trepalin, S. V. (1999). HYBOT program package. Registration by Russian State Patent Agency, (990090).
- Trepalin, S.V., Yarkov, A.V. (2001). CheD: Chemical Database Compilation Tool, Internet Server, and client for SQL Servers. J. Chem. Inf. Comput. Sci., 41, 100-107. DOI
- Verma, J., Khedkar, V. M., & Coutinho, E. C. (2010). 3D-QSAR in drug design-a review. Current topics in medicinal chemistry, 10(1), 95-115. DOI
- SYBYL-Х 2.1. Certara, Princeton, NJ, USA
- Pearlman, D. A., Case, D. A., Caldwell, J. W., Seibel, G. L., Singh, U. C., Weiner, P., & Kollman, P. A. (1991). AMBER 4.0, University of California, San Francisco.
- Boobbyer, D. N., Goodford, P. J., McWhinnie, P. M., & Wade, R. C. (1989). New hydrogen-bond potentials for use in determining energetically favorable binding sites on molecules of known structure. Journal of medicinal chemistry, 32(5), 1083-1094. DOI
- Groom, C. R., Bruno, I. J., Lightfoot, M. P., & Ward, S. C. (2016). The Cambridge structural database. Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials, 72(2), 171-179. DOI
- Raevsky, O. A., & Skvortsov, V. S. 3D HYBOT programm. Russian State Patent Department, (004612207).
- Trepalin, S. V., Yarkov, A. V., & Raevsky, O. A. (2018). MOLTRA-II. New three dimensional descriptors of the hydrogen bond. Biomedical Chemistry: Research and Methods, 1(3), e00069. DOI
- Van de Waterbeemd, H., & Testa, B. (1987). The parametrization of lipophilicity and other structural properties in drug design. Advances in drug research, 16, 85-225.
- Raevsky, O. A., Schaper, K. J., & Seydel, J. K. (1995). H‐Bond Contribution to Octanol‐Water Partition Coefficients of Polar Compounds. Quantitative Structure‐Activity Relationships, 14(5), 433-436. DOI
- Raevsky O.A., Trepalina E.P., Trepalin S.V.(2000) in Molecular Modelling and Prediction of Bioactivity (Gundertofe K. and Jorgensen F.eds.), Kluwer Academic/Plenum Publ.; p. 489-490.
- Raevsky, O. A. (2001). Molecular lipophilicity calculations of chemically heterogeneous chemicals and drugs on the basis of structural similarity and physicochemical parameters. SAR and QSAR in Environmental Research, 12(4), 367-381. DOI
- Raevsky, O. A., Trepalin, S. V., Trepalina, H. P., Gerasimenko, V. A., & Raevskaja, O. E. (2002). 41 SLIPPER-2001− Software for predicting molecular properties on the basis of physicochemical descriptors and structural similarity. Journal of chemical information and computer sciences, 42(3), 540-549. DOI
- Lipinski, C. A. (2000). Drug-like properties and the causes of poor solubility and poor permeability. Journal of pharmacological and toxicological methods, 44(1), 235-249. DOI
- Alelyunas, Y. W., Empfield, J. R., McCarthy, D., Spreen, R. C., Bui, K., Pelosi-Kilby, L., & Shen, C. (2010). Experimental solubility profiling of marketed CNS drugs, exploring solubility limit of CNS discovery candidate. Bioorganic & medicinal chemistry letters, 20(24), 7312-7316. DOI
- Hansch, C., Quinlan, J. E., & Lawrence, G. L. (1968). Linear free-energy relationship between partition coefficients and the aqueous solubility of organic liquids. The Journal of Organic Chemistry, 33(1), 347-350. DOI
- Schaper, K. J., Kunz, B., & Raevsky, O. A. (2003). Analysis of water solubility data on the basis of HYBOT descriptors: Part 2. Solubility of liquid chemicals and drugs. QSAR & Combinatorial Science, 22(9‐10), 943-958. DOI
- Dearden, J. C. (2006). In silico prediction of aqueous solubility. Expert opinion on drug discovery, 1(1), 31-52. DOI
- Faller, B., & Ertl, P. (2007). Computational approaches to determine drug solubility. Advanced drug delivery reviews, 59(7), 533-545. DOI
- Johnson, S. R., & Zheng, W. (2006). Recent progress in the computational prediction of aqueous solubility and absorption. The AAPS journal, 8(1), E27-E40. DOI
- Sugano, K., Okazaki, A., Sugimoto, S., Tavornvipas, S., & Omura, A. (2007). Solubility and dissolution profile assessment in drug discovery. Drug metabolism and pharmacokinetics, 22(4), 225-254. DOI
- Wang, J., & Hou, T. (2011). Recent advances on aqueous solubility prediction. Combinatorial chemistry & high throughput screening, 14(5), 328-338.
- Skyner, R. E., McDonagh, J. L., Groom, C. R., Van Mourik, T., & Mitchell, J. B. O. (2015). A review of methods for the calculation of solution free energies and the modelling of systems in solution. Physical Chemistry Chemical Physics, 17(9), 6174-6191. DOI
- Chevillard, F., Lagorce, D., Reynès, C., Villoutreix, B. O., Vayer, P., & Miteva, M. A. (2012). In silico prediction of aqueous solubility: a multimodel protocol based on chemical similarity. Molecular pharmaceutics, 9(11), 3127-3135. DOI
- Yalkowsky, S. H., & Valvani, S. C. (1980). Solubility and partitioning I: solubility of nonelectrolytes in water. Journal of pharmaceutical sciences, 69(8), 912-922. DOI
- Raevsky, O. A., Polianczyk, D. E., Grigorev, V. Y., Raevskaja, O. E., & Dearden, J. C. (2015). In silico prediction of aqueous solubility: A comparative study of local and global predictive models. Molecular informatics, 34(6‐7), 417-430. DOI
- Raevsky, O. A., Grigor’ev, V. Y., Polianczyk, D. E., Raevskaja, O. E., & Dearden, J. C. (2014). Calculation of aqueous solubility of crystalline un-ionized organic chemicals and drugs based on structural similarity and physicochemical descriptors. Journal of chemical information and modeling, 54(2), 683-691. DOI
- DRAGON, version 5.5;Talete srl, Milano, Italy, 2011.
- Obrezanova, O., Csányi, G., Gola, J. M., & Segall, M. D. (2007). Gaussian processes: a method for automatic QSAR modeling of ADME properties. Journal of chemical information and modeling, 47(5), 1847-1857. DOI
- Obrezanova, O., Gola, J. M., Champness, E. J., & Segall, M. D. (2008). Automatic QSAR modeling of ADME properties: blood–brain barrier penetration and aqueous solubility. Journal of computer-aided molecular design, 22(6-7), 431-440. DOI
- Raevsky, O. A., Grigorev, V. Y., Polianczyk, D. E., Raevskaja, O. E., & Dearden, J. C. (2017). Six global and local QSPR models of aqueous solubility at pH= 7.4 based on structural similarity and physicochemical descriptors. SAR and QSAR in Environmental Research, 28(8), 661-676. DOI
- Raevsky, O. A., Fetisov, V. I., Trepalina, E. P., McFarland, J. W., & Schaper, K. J. (2000). Quantitative estimation of drug absorption in humans for passively transported compounds on the basis of their physico‐chemical parameters. Quantitative Structure‐Activity Relationships, 19(4), 366-374. DOI
- Raevsky, O. A., & Skvortsov, V. S. (2005). Quantifying hydrogen bonding in QSAR and molecular modeling. SAR and QSAR in Environmental Research, 16(3), 287-300. DOI
- Raevsky, O. A., Schaper, K. J., Artursson, P., & McFarland, J. W. (2001). A novel approach for prediction of intestinal absorption of drugs in humans based on hydrogen bond descriptors and structural similarity. Quantitative Structure‐Activity Relationships, 20(5‐6), 402-413. DOI
- Bradbury, M. W. B. (1979). The concept of a blood-brain barrier. John Wiley & Sons.
- Wolf, S., Seehaus, B., Minol, K., & Gassen, H. G. (1996). The blood-brain barrier: a specialty of cerebral microcirculation systems. Die Naturwissenschaften, 83(7), 302-311.
- Pauletti, G. M., Okumu, F. W., & Borchardt, R. T. (1997). Effect of size and charge on the passive diffusion of peptides across Caco-2 cell monolayers via the paracellular pathway. Pharmaceutical research, 14(2), 164-168. DOI
- Hansch, C., Steward, A. R., Anderson, S. M., & Bentley, D. L. (1968). Parabolic dependence of drug action upon lipophilic character as revealed by a study of hypnotics. Journal of medicinal chemistry, 11(1), 1-11. DOI
- Young, R. C., Mitchell, R. C., Brown, T. H., Ganellin, C. R., Griffiths, R., Jones, M., ... & Smith, I. R. (1988). Development of a new physicochemical model for brain penetration and its application to the design of centrally acting H2 receptor histamine antagonists. Journal of medicinal chemistry, 31(3), 656-671. DOI
- van de Waterbeemd, H., & Kansy, M. (1992). Hydrogen-bonding capacity and brain penetration. CHIMIA International Journal for Chemistry, 46(7-8), 299-303.
- Lipinski, C. A., Lombardo, F., Dominy, B. W., & Feeney, P. J. (1997). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced drug delivery reviews, 23(1-3), 3-25. DOI
- Norinder, U., & Haeberlein, M. (2002). Computational approaches to the prediction of the blood–brain distribution. Advanced drug delivery reviews, 54(3), 291-313. DOI
- Didziapetris, R., Japertas, P., Avdeef, A., & Petrauskas, A. (2003). Classification analysis of P-glycoprotein substrate specificity. Journal of drug targeting, 11(7), 391-406. DOI
- Pajouhesh, H., & Lenz, G. R. (2005). Medicinal chemical properties of successful central nervous system drugs. NeuroRx, 2(4), 541-553. DOI
- Borchardt, R., Kerns, E., Hageman, M., Thakker, D., & Stevens, J. (Eds.). (2007). Optimizing the" drug-like" Properties of Leads in Drug Discovery. Springer Science & Business Media.
- Wager, T. T., Chandrasekaran, R. Y., Hou, X., Troutman, M. D., Verhoest, P. R., Villalobos, A., & Will, Y. (2010). Defining desirable central nervous system drug space through the alignment of molecular properties, in vitro ADME, and safety attributes. ACS chemical neuroscience, 1(6), 420-434. DOI
- Ghose, A. K., Herbertz, T., Hudkins, R. L., Dorsey, B. D., & Mallamo, J. P. (2011). Knowledge-based, central nervous system (CNS) lead selection and lead optimization for CNS drug discovery. ACS chemical neuroscience, 3(1), 50-68. DOI
- Desai, P. V., Sawada, G. A., Watson, I. A., & Raub, T. J. (2013). Integration of in silico and in vitro tools for scaffold optimization during drug discovery: predicting P-glycoprotein efflux. Molecular pharmaceutics, 10(4), 1249-1261. DOI
- Panarin, V. A., Kondratyev, V. A., & Rayevsky, O. A. (1990). Some characteristics of the functioning of membrane receptor‐channel complexes of Limnaea stagnalis neurones. The Journal of physiology, 423(1), 363-380. DOI
- Hitchcock, S. A., & Pennington, L. D. (2006). Structure− brain exposure relationships. Journal of medicinal chemistry, 49(26), 7559-7583. DOI
- Raevsky, O. A. (1990). The structure and properties of complexes simulating molecular recognition. Uspekhi Khimii, 59(3), 375-400.
- Raevsky, O. A. (2004). Physicochemical descriptors in property-based drug design. Mini reviews in medicinal chemistry, 4(10), 1041-1052. DOI
- van de Waterbeemd, H., Camenisch, G., Folkers, G., Chretien, J. R., & Raevsky, O. A. (1998). Estimation of blood-brain barrier crossing of drugs using molecular size and shape, and H-bonding descriptors. Journal of drug targeting, 6(2), 151-165. DOI
- Raevsky, O. A., & Schaper, K. J. (1998). Quantitative estimation of hydrogen bond contribution to permeability and absorption processes of some chemicals and drugs. European journal of medicinal chemistry, 33(10), 799-807. DOI
- Raevsky, O. A., Solodova, S. L., Raevskaya, O. E., Liplavskiy, Y. V., & Mannhold, R. (2012). The computer classification models on the relationship between chemical structures of compounds and drugs with their blood brain barrier penetration ability. Biochemistry (Moscow) Supplement Series B: Biomedical Chemistry, 6(1), 31-38. DOI
- Raevsky, O. A., Solodova, S. L., Raevskaya, O. E., & Mannhold, R. (2012). Quantitative interaction between the structures of organic compounds and their abilities to penetrate the blood-brain barrier. Pharmaceutical Chemistry Journal, 46(3), 133-138. DOI
- Rankovic, Z. (2015). CNS drug design: balancing physicochemical properties for optimal brain exposure. Journal of medicinal chemistry, 58(6), 2584-2608. DOI
- Raevsky, O. A. (2016). CNS Multiparameter Optimization Approach: Is it in Accordance with Occam’s Razor Principle?. Molecular informatics, 35(3‐4), 94-98. DOI
- Raevsky, O. A., Polianczyk, D. E., Mukhametov, A., & Grigorev, V. Y. (2016). Assessment of the classification abilities of the CNS multi-parametric optimization approach by the method of logistic regression. SAR and QSAR in Environmental Research, 27(8), 629-635. DOI
- Raevsky, O. A., Grigorev, V. Y., Polianczyk, D. E., Sandakov, G. I., Solodova, S. L., Yarkov, A. V., Bachurin, S.O. & Dearden, J. C. (2016). Physicochemical property profile for brain permeability: comparative study by different approaches. Journal of drug targeting, 24(7), 655-662. DOI