|
CONTENTS 1. BACTERIAL "FRANKENSTEIN": HOW TO TURN AN EVIL MICROBE INTO A GOOD DOCTOR 2. THE BACTERIAL ROBOT ENTERS THE GAME: MANAGEMENT AND DELIVERY 3. HEAVILY ARMED: THE THERAPEUTIC AND DIAGNOSTIC POTENTIAL OF THE BACTERIAL ROBOT 5. BACTERIAL ROBOT... WITHOUT BACTERIA: THE PROSPECTS COMPLIANCE WITH ETHICAL STANDARDS Figure 5Invasion of a bacterium in a cancer cell to release a therapeutic cargo Table 1Various bacterial modification strategies Table 1Principles of development of a bacterial robot for cancer therapy Table 1Biological carriers in the creation of a bacterial robot |
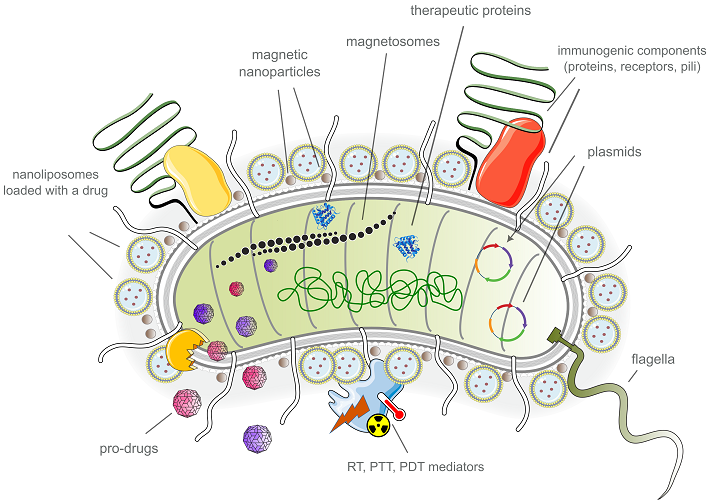
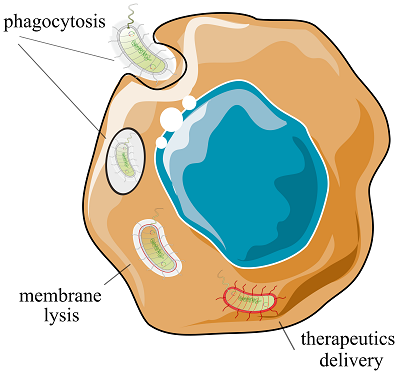
BACTERIOBOTS: BACTERIAL MICROROBOTS AND THEIR POTENTIAL IN CANCER DIAGNOSIS AND THERAPY Sechenov First Moscow State Medical University, 8/2 Trubetskaya str., Moscow, 119991 Russia; e-mail: larisa.ikryannikova@gmail.com Key words: bacterial microrobot; bacteriobot; bacterial biohybrid; antitumor therapy DOI: 10.18097/BMCRM00301 INTRODUCTION Cancer is one of the leading causes of death worldwide, claiming more than 10 million lives annually. While significant efforts have been made to develop ways to fight cancer, current cancer treatments still face several limitations, including serious side effects, limited efficacy against metastasis and recurrence, and high costs for each patient. Therefore, the development of novel therapeutic approaches in cancer research is of utmost importance [1]. The concept of a "bacteriobot", a bacterial microrobot, emerged in scientific circles relatively recently. This term was probably first introduced in 2013-2014, to denote a new innovative therapeutic method for manufacturing bacterial-based drugs for treatment of malignant tumors [2-3]. The idea of using bacteria as therapeutic agents is based on their versatility and plasticity as a natural biomaterial. Bacteria can move independently and under certain conditions penetrate into hard-to-reach tumor tissues, making them a suitable delivery system for therapeutic agents. Additionally, bacteria have natural immunogenic properties that can enhance the effectiveness of cancer immunotherapy [4-6]. A bacteriobot is a construct that combines a microsensor for detecting malignant cells, a microactuator for reaching a target, and a therapeutic agent for treatment [2]. A bacteriobot can be defined as any bacterium that has been modified in some way, for example through genetic engineering. However, the current scientific literature suggests a narrower definition, which describes a bacterial robot as an object that combines natural or genetically modified bacteria with synthetic materials and structures on a nanoscale or microscale [7-12]. The history of using bacteria in antitumor therapy dates back to ancient times. More than three thousand years ago, the Egyptian physician Imhotep documented the relationship between bacterial infections and the treatment of "tumors", which may have been cancerous tissues [13-14]. In medical documents from the first third of the 19th century, observations describing the fact that malignant tumors regressed in people exposed to bacterial infections were mentioned [15]. The case report described by the German physician W. Busch in the 1860s, which became widely known and included into textbooks, involved a patient with an unknown cause of cancer who was intentionally infected. The patient was placed in a bed that had recently been occupied by another patient who had died from erysipelas, a disease caused by Streptococcus pyogenes. As expected, infection occurred, and the tumor actually began to regress, but the patient soon died from streptococcal infection [13, 16-17]. Later, the so-called infectious theory of carcinogenesis emerged, linking the formation and spread of tumors to bacterial or parasitic infections. During this time, in the late 19th and early 20th centuries, researchers and physicians were trying to find the "cancer germ", a microbial agent that caused cancer, and to prove its role in cancer formation [18-21]. At the same time, efforts were made to develop the first anticancer vaccines based on inactivated or weakened bacteria. One of the most remarkable examples is a drug known as "Coley's toxins". This drug was created in the early 20th century by the surgeon W. Coley. He noticed tumor regression in patients with bone sarcoma when the wound surface was infected after surgery. The drug was a mixture of heat-inactivated bacteria, S. pyogenes and Serratia marcescens [22-24]. Researchers often tried to develop antitumor vaccines using microorganisms that they observe (or believe they observe) microscopically in tumor tissue samples or the blood of cancer patients [16, 18, 25-26]. Despite considerable efforts and some successes, a universal, effective, and reliable vaccine against cancer based on microorganisms has not been developed. As a result, the theory of bacterial (or parasitic) oncogenesis, that was popular at the turn of the 19th and 20th centuries, was generally rejected by the scientific and medical communities by the middle of the last century, and almost forgotten. The information accumulated over more than a hundred years remained largely unclaimed when two significant events occurred in the scientific world at the end of the 20th century. Firstly, the etiological role of Helicobacter pylori in the development of gastric cancer was established or, more precisely, officially recognized [27]. In addition to H. pylori, there are other bacteria that have been linked to individual cancer processes, although they are not yet officially recognized as the main cause. For example, Bacteroides fragilis, Streptococcus bovis, and Fusobacterium nucleatum have been strongly associated with colorectal cancer. Chlamydia trachomatis has been linked to cervical cancer, while Porphyromonas gingivalis is associated with oral cancer [28-30]. Secondly, there has been a significant qualitative leap in the development of molecular biological tools. This, among other things, helps to investigate the taxonomic composition of microbial communities without the need for cultivating pure bacterial cultures. This has brought us closer to understanding the "microbiome" of malignant tumors and its possible role in cancer development. The revival of the idea of using microbes to fight cancer has also been facilitated by the rapid development of genetic engineering techniques, particularly genome editing methods. A hundred years ago researchers tried to create the first antitumor "vaccines" based on natural, native bacteria, at best devoid of virulence properties by repeatedly passaging the strain. But the modern "bacterial" anticancer drugs are truly masterpiece of genetic engineering, where virulence factors are removed from the bacterial cell, while immunogenicity properties are enhanced and the specificity to tumor cells are significantly increased. Some bacterial phyla, such as Clostridia, Salmonella or Listeria, attract special attention of researchers because these bacteria are anaerobic and have the ability to invade and survive in hypoxic areas inside solid tumors. Drugs developed on the basis of genetically modified strains of these bacteria are currently at various stages of clinical research [31-35]. The last and most significant step towards creating bacterial robots was the development of technologies for working with micro- and nano-scale objects. The combination of nanotechnology with biological carriers seems to be the most promising approach for overcoming physiological barriers, such as the blood-brain barrier and hypoxic foci in tumors, and for targeted delivery of therapeutic agents [36]. Chemically, physically, and biologically modified bacteria that are invisible to the human immune system when outside the tumor become real "police robots" in the fight against cancer. These bacteria are armed with therapeutic agents that target cancer cells and protect human health. This review focuses on the ways in which these bacterial robots are created, targeted, and supplied with therapeutics. 1. BACTERIAL "FRANKENSTEIN": HOW TO TURN AN EVIL MICROBE INTO A GOOD DOCTOR A bacteriobot is a tiny device based on bacteria or parts of bacteria. It combines the functions of a sensor that detects a target, a therapeutic agent or container for transferring therapeutics, and an actuator, which directs the device to its destination and starts its operation (Fig. 1) [37]. This device can be created using biological, chemical, or physical methods [6, 38-40]. It may contain only cellular components, or it may be a biohybrid of cellular and synthetic parts. The last ones should be biocompatible and biodegradable, making them safe for use in the body.
Biological methods primarily refer to methods of genetic engineering in bacteria. The rapid development of genetic technology in recent decades, particularly high-throughput sequencing platforms, has led to an accumulation of information about bacterial genome structure. This, in turn, has enabled identification of a large number of functional genetic elements, creating a basis for the design of genetically modified bacteria. These modified bacteria have the ability to produce therapeutic drugs directly inside a tumor or enhance the immune response. A significant part of genetically engineered modifications aims to reduce bacterial virulence for patient safety and increase tumor tropicity [10]. There are many examples of genetic modifications of bacteria for oncotherapy, but we will not go into details in this article and address readers to some excellent reviews [11-12, 34, 41-44] and examples that can be found in Table 1.
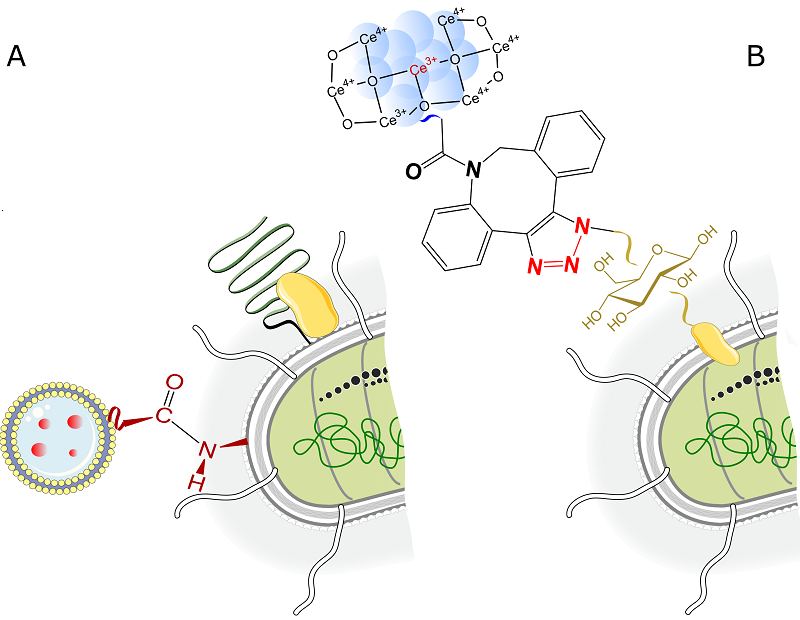
Chemical modification methods involve the targeted attachment of various types of biocompatible and biodegradable components to bacteria through the creation of chemical crosslinks. The bacterial cell wall is composed of peptidoglycan, a polysaccharide structure formed by N-acetylmuramic acid and N-acetylglucosamine through β-1,4 glycoside bonds. This surface, which is rich in amino and carboxyl groups, serves as a reaction site for chemical cross-linking. The work by Taherkhani et al. [49] provides an example of the covalent binding of Magnetococcus marinus bacteria with drug-loaded nanoliposomes to create a magnetotactic bacterial structure that can move with the help of an external magnetic field. This bioconjugation is based on the carbodiimide method, which forms an amide (peptide) bond between activated carboxyl groups on the nanoliposome surface and amino groups on the bacterial cell wall of M. marinus (Figure 2A). Similarly, the amide bond formation between carboxylated magnetic nanoparticles (MNP-COOH) and the surface of a genetically modified probiotic strain of Escherichia coli Nissle 1917 resulted in the creation of the EcN@MNP biohybrid, which is sensitive to a magnetic field and is suitable for further modification [55]. The principles of carbodiimide chemistry have been successfully applied to create a biohybrid complex based on the non-pathogenic E. coli MG1655 strain, which produces large amounts of cytotoxic protein ClyA and bismuth sulfide (III) nanoparticles [56]. Bi2S3 (BNP) nanoparticles are bound to the bacterial cell wall by a peptide fragment that mimics a human matrix metalloproteinase-2 (MMP-2) substrate. The colonization of tumors with this biohybrid E. coli@BNP was accompanied by the degradation of the peptide fragment by MMP-2, present in the tumor matrix. This lead to the release of BNP nanoparticles, which could increase sensitivity to ionizing radiation during radiotherapy due to the generation of reactive oxygen species (ROS) [56]. The streptavidin-biotin complex is well-known tool in the arsenal of biotechnologists to form strong and stable non-covalent bonds between fragments of biohybrid structures. Liposomes loaded with the therapeutic agent paclitaxel have been successfully sewn to Salmonella typhimurium strains by binding streptavidin, which covers the liposome surface, to biotin on the outer membrane proteins of the bacterium [50]. In a similar manner, a hybrid microplatform has been created using the biotin-streptavidin crosslinking that consists of a genetically modified strain of E. coli MG1655 strain, magnetic nanoparticles for motion control, and nanoliposomes loaded with doxorubicin and the photothermal agent indocyanine green [57]. The bioorthogonal reaction of an azide-cyclooctin pair should be mentioned when speaking about the methods of chemical engineering of bacteria. This is a complex and wonderful approach proposed by Pan [51] to create a bacterial-abiotic hybrid of E. coli with cerium (IV) oxide nanoparticles. The term "bioorthogonal reaction" refers to chemical processes that occur inside cells or living organisms under physiological conditions without affecting the natural biochemical processes. The substrate, 2-azido-2-deoxy-D-glucose, was selected as a sugar source for bacterial cultivation. It was used to produce azide on the cell wall of the E. coli strain MG1655 (N3-Bac). Cerium oxide was then modified with azo-dibenzocyclooctin (DBCO) to form the СеО2-DBCO complex. Finally, the cerium oxide nanoparticles were anchored to the surface of the bacterium through a bioorthogonal click reaction between the -DBCO and -N3 fragments (Fig. 2B). A modified bacterium has been designed to simultaneously absorb lead cations and neutralize ROS in the human tissues. The similar method was in [58] to create a chemical bond between DBCO-modified paramagnetic Fe3O4 nanoparticles and a genetically engineered avirulent E. coli BL21 strain.
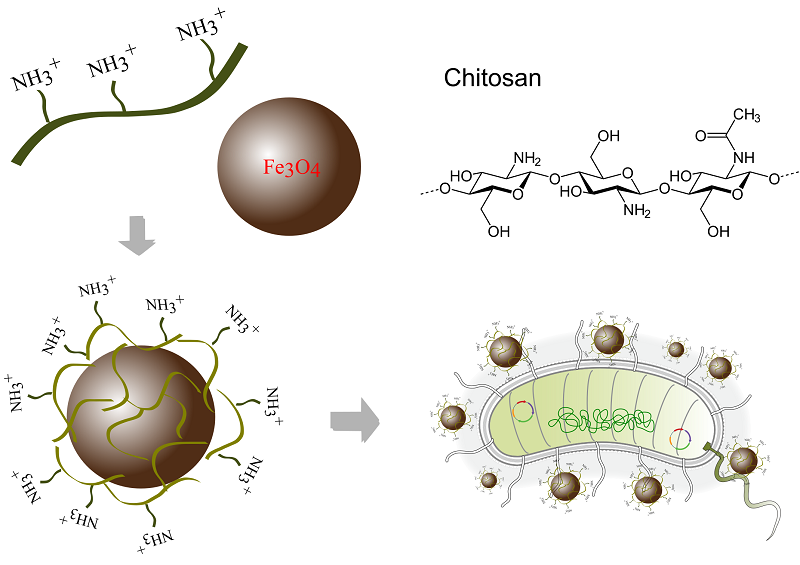
Another example of chemical modification of bacteria is the creation of bacterial-inorganic composites based on natural biomineralization processes [59]. Biomineralization is the process of formation of solid inorganic substances inside a living organism. Magnetotactic bacteria, for example, synthesize magnetic nanoparticles in their cytoplasm, making them sensitive to external magnetic fields. This property can be harnessed for imaging tumors and delivering therapeutic agents [60]. The ability to biomineralize offers promising possibilities for creating biohybrids with unique properties. A hybrid photothermally activated bacteria based on E. coli MG1655 strain was developed by Wang et al. [61]. The bacteria were transformed with a plasmid containing the gene of the cytotoxic pore-forming protein, cytolysin A (ClyA), with a thermosensitive promoter enabling the activation of clyA expression through thermal exposure. Next, the bacteria were cultivated together with chloroauric acid H[AuCl4]. During cultivation, H[AuCl4] was reduced on the bacterial cell surface to form gold nanoparticles under the action of bacterial NADPH reductase, and this resulted in the formation of thermally activated biohybrid (TAB) - TAB@Au. Under the influence of laser irradiation, the gold nanoparticles were heated, activating the expression of the cytolysin gene and destroying tumor cells [61]. Organometallic compounds, such as zeolite-like imidazolate frameworks (ZIF) with high sorption capacity, are used to create bacteria-based biomimetic materials. Such approach was employed in [62]; the authors used organometallic ZIF-90 imidazolate framework loaded with methylene blue, which was attached to the surface of the anaerobic bacterium Shewanella oneidensis MR-1. This was achieved by forming an imine group through the reaction between the amino group on the bacterial surface and the aldehyde group on the organometallic compound. Methylene blue, trapped inside the ZIF-90 structure, generates singlet oxygen (1О2) in illuminated tumor cells, thus causing mitochondrial dysfunction, inhibition of ATP synthesis, and suppression of gene expression of heat shock proteins. Yan et al. [53] constructed a composite using the E. coli strain MG1655 along with an imidazolate framework ZIF-8, which was loaded with a photosensitizer (Chlorin E6) and a cytostatic drug, doxorubicin. This composite was designed for the combined treatment of tumor cells. Biohybrid E.coli@ZIF-8/C&D was created through a chemical reaction between 2-methylimidazole and zinc (II) sulfate, in the presence of E. coli, Chlorin E6 (C) and doxorubicin (D), resulting in ZIF-8 particles being evenly distributed on the surface of the bacterial cell, with the photosensitizer and therapeutic agent trapped within them [53]. Among the physical methods of bacterial modification, electrostatic interaction is the most commonly used. Under normal physiological conditions, the bacterial cell wall has a small negative charge, which allows for stable electrostatic contact with positively charged nanoparticles, such as gold nanoparticles functionalized with a cationic polyethylenimine with a branched structure [63]. It has been shown that the strength of the binding between gold nanoparticles and the surface of a Gram-positive Bacillus subtilis strain depends on the availability of negative charged teichoic acid fragments on the bacterial cell wall [63]. Chitosan, a polycationic biopolymer with a large number of amino groups, has been proposed as a method to modify magnetic iron oxide nanoparticles [64]. These modified nanoparticles have been shown to effectively bind to the walls of both Gram-negative E. coli and Gram-positive Staphylococcus aureus (Fig. 3).
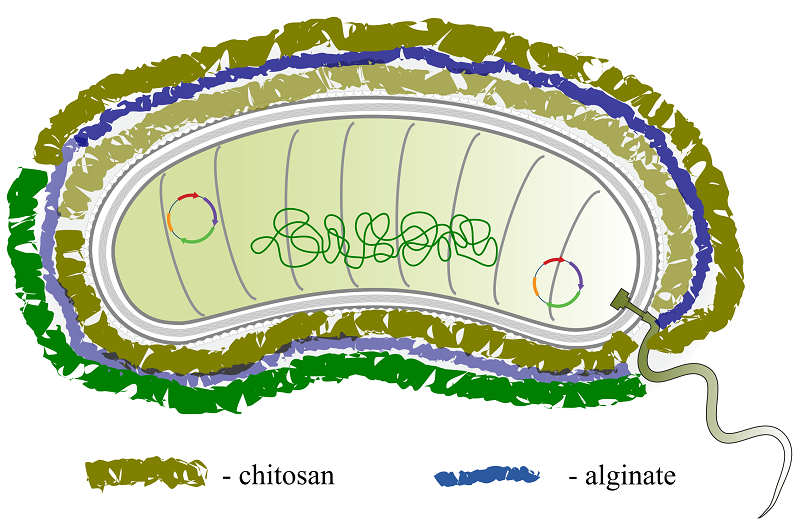
The strategy of using a nanophotocatalyst to «charge» bacteria and enhance their metabolic activity was proposed by Zheng et al. [65]. E. coli MG1655 cells were coated electrostatically with carbon nitride (C3N4) doped with carbon particles (carbon-dot doped carbon nitride, CCN). The resulting construct, CCN@E.coli, was proposed for photoregulated therapy with involvement of bacterial metabolites, where light exposure stimulated of photoelectrons transfer from the C3N4 to the bacteria, followed by the enzymatic conversion of endogenous nitrate anions to cytotoxic nitric oxide (NO) in the area of a malignant neoplasm. In a study by Ding et al. [66], ultra-small (3 nm) particles of black phosphorus (BPQDs, black phosphorus quantum dots) were electrostatically adsorbed onto the surface of E. coli Nissle 1917 strain. This strain had been previously genetically modified to increase the production of catalase. BPQDs are characterized by their ability to absorb light energy over a wide range of wavelengths and have high photothermal conversion efficiency. When laser irradiation is applied to the E.coli/BPQDs biohybrid at a wavelength of 660 nm, it generates ROS damaging the bacterial cell membrane; and released catalase cleaving hydrogen peroxide, also generates ROS. As a result, the E.coli/BPQDs construct maintains a high level of ROS in the vicinity of the malignant tumor, enhancing the overall effectiveness of photodynamic therapy. However, it should be noted that bacterial biohybrids formed by means of the electrostatic interactions may not to be sufficiently durable for the in vivo use. To some extent, this issue can be addressed by a strategy of layer-by-layer self-assembly of modified components on the surface of bacteria. For example, in [54], a probiotic strain of Bacillus coagulans was successfully encapsulated within a shell of two biodegradable polysaccharides, chitosan and alginate, to protect it in the intestine. Alternating layers of cationic chitosan and anionic alginate were sequentially deposited on the surface of B. coagulans by electrostatic forces, forming a shell up to three bilayers thick (Fig. 4). An interesting solution was proposed in [67], where nanosheets of layered double cobalt-copper-molybdenum hydroxides (CoCuMo-LDH, layered double hydroxides) were firmly attached to the bacterial membrane of the probiotic strain Lactobacillus acidophilus. Subsequently, with the involvement of lactobacillus metabolites, the crystalline nanosheets underwent a phase transformation to an amorphous state, which was characterized by improved photodynamic properties.
2. THE BACTERIAL ROBOT ENTERS THE GAME: MANAGEMENT AND DELIVERY Effective drug delivery to areas of malignancy and metastasis is a major challenge in cancer treatment. This is particularly true for solid tumors located in difficult-to-access areas of the body, where passive intravenous delivery is inefficient and intracellular transport is limited. Compared to normal tissue, the tumor's vascular system is disorganized, with a large distance between capillaries, so only a small percentage (often less than 1%) of intravenously administered drugs enter the tumor [68-69]. It is known that therapeutic agents with a size less than ~5 nm (all low molecular weight drugs) are subject to renal filtration and clearance. This requires higher start drug concentrations and is associated with an increased risk of side effects. On the other hand, therapeutic agents greater than 100 nm cannot easily penetrate the tiny pores in the vascular network [4]. The problem of ineffective drug delivery is further complicated by rapid cancer cell growth and the presence of a dense extracellular matrix. This, combined with a lack of lymphatic drainage, makes it even more difficult for drugs to be transported. A miniature robot equipped with a therapeutic agent and capable of moving independently towards and inside a tumor could help solve this problem. The main advantage of living bacteria, which are the cornerstone of such a robot, is their mobility, which allows them to actively leave the vascular system and enter tumor tissues [70]. Another important feature of bacteria is their ability to survive and function independently in severe physiological conditions for some time. However, it is not an easy task for a bacterium to reach a tumor. On a long journey through the body, they must avoid immune attacks and overcome various obstacles before entering the tumor and releasing the therapeutic agent (see Table 2).
2.1. Moving towards the tumor.
It is well known that certain types of anaerobic microorganisms, such as strictly anaerobic clostridia, bifidobacteria, and facultatively anaerobic bacteria like salmonella, listeria, and E. coli, can enter the bloodstream and reach tumors, selectively colonizing their hypoxic and necrotic areas [71]. It was shown in one early study, that infection of melanoma-bearing mice with an intraperitoneal injection of the wild-type strain of S. typhimurium (ATCC 14028) led to the accumulation of bacteria within the tumor at the levels exceeding 109 colony-forming units (CFUs) per gram of tumor tissue [72]. An additional factor that attracts bacteria to the tumor area is the immunodeficient microenvironment of the neoplasm. This helps microorganisms avoid attacks from the immune system. Additionally, the presence of chemotactic tissue breakdown products contributes to this process [73]. The movement of bacteria through the bloodstream can occur passively, with gradual accumulation in tumor area, or actively, for example, using flagella. Bacterial flagella rotate like propellers and can help bacteria move at speeds of up to 300 µm/s [4]. There was a reason to believe that the delivery of a therapeutic cargo by bacteria would not significantly alter the physiological characteristics of the microorganism or speed of its preventing the movement of biohybrids and their penetration into tumor foci. Indeed, at least at the level of cellular models [74], including spatial ones [75], it has been demonstrated that the conjugation of nanoscale container particles with S. typhimurium does not interfere with the intracellular transport of the bacterium. At the same time, the concentration of the drug delivered by a bacteriobot is approximately 100 times higher than the amount of therapeutic cargo that would penetrate the tumor through passive diffusion [75]. In an isotropic environment, without attractants, bacteria move randomly and chaotically. As mentioned above, chemotaxis towards the areas of necrosis in tumor tissues provides a some attraction for the movement of bacteria, and consequently, bacteriobots [37]. The idea of using external forces to direct the movement of a bacterial robot seems promising. A popular and rapidly growing approach is the use of magnetic fields to guide bacteriobots from the injection site into the body to the tumor localization site [76, 77]. In this case, the bacteria or bacterial robot would need to be sensitive to magnetic fields. For example, the magnetotactic Magnetococcus marinus (MC-1), a gram-negative coccus that can be found in the Atlantic Ocean, is an example of such a bacterium [52]. The magnetosomes of this bacterium are magnetite particles (Fe3O4), surrounded by membranes and forming chains in the cytoplasm. Control by using an external magnetic field of an MC-1 biohybrid conjugated with drug-loaded nanoliposomes when injected peritumorally into live mice, leads to a significant penetration (up to 55%) of MC-1 into the hypoxic regions of colorectal tumors. It should be noted that there were no obvious signs of fever or toxicity in mice due to the introduction of a foreign microorganism [78]. In addition to M. marinus, at least several other species of naturally magnetotropic bacteria are under active development, including Magnetovibrio MV-1 and members of the genus Magnetospirillum – M. gryphiswaldense MSR-1, M. magneticum AMB-1 and M. magnetotacticum MS-1 [76]. It is also possible to impart the magnetosensitivity to a bacterium that did not originally possess this property. Aubry et al. [79] created the E. coli strain that stimulated the formation of iron-rich structures inside the cell due to the overexpression of the ferritin protein gene during growth in a medium enriched with ferruginous compounds. Other researchers [57] have shown that the sensitivity of the E. coli based biohybrid to external magnetic fields is achieved by attaching magnetic nanoparticles to the bacterial cell wall. This biohybrid structure also included nanoliposomes loaded with doxorubicin and a photothermal agent, and was able to move under the influence of a magnetic field within biological matrices. It can effectively colonized tumor spheroids and released a therapeutic agent when exposed to infrared radiation. A "hybrid" microrobot was created based on an attenuated strain of S. typhimurium in [80]. The robot consisted of magnetic nanoparticles (Fe3O4), which were enclosed in a biodegradable alginate-chitosan capsule and electrostatically attached to the surface of the bacterial cell wall of salmonella. The tests in a gel microfluidic system confirmed that the biohybrid was able to move under the influence of an external electromagnetic field, even against the flow of blood in large vessels. However, in smaller vessels and within tissues, movement was controlled by the activity of bacterial flagella and the chemotaxis. 2.2. Targeting and DetectionThe movement of the bacterial robot towards the tumor area can be artificially stimulated. Typically, the increased affinity of a bacteriobot for a tumor is associated with genetic modification of its core element, the bacterium. Due to mutations and other genetic or epigenetic abnormalities, cancer cells and their microenvironment acquire chemical and physical characteristics that differ from those of healthy cells. In particular, cancer cells produce abnormal cell surface proteins called neoantigens, which are unique to the tumor and therefore serve as specific targets [81]. Genetic modification of a bacterium can help bring tumor-specific ligands to its surface. Park et al. [82] developed a ΔppGpp-deficient strain of Salmonella typhimurium (SHJ2037) with the ability to display the RGD peptide fragment (ACDCRGDCFCG) on the outer membrane protein OmpA. The RGD peptide binds to the avβ3 integrin receptor, which is overexpressed by cancer cells. This allows the bacterial strain to be specifically targeted at tumors. The strain SHJ2037 showed high tumor specificity and a significant therapeutic effect in breast cancer and skin cancer models. The tumor specificity of salmonella can be enhanced by synthesizing a single-domain antibody (VHH) on the bacterial surface that targets the tumor-associated antigen hCD20 [83]. This antibody-dependent targeting determines the effectiveness of the infection of CD20+ lymphoma cells both in vitro and in vivo, with a significant reduction in non-specific cell invasion. Another approach is the use of synthetic adhesines on the bacterial cell wall; this increases the specificity of bacterial adhesion to cancer cells [71]. These engineered adhesines have a modular structure: the cylindrical beta domain of the intimin surface protein is anchored in the bacterial membrane and holds an immunoglobulin-like (Ig) adhesive VHH domain. This structure can be easily modified to any type of tumor by replacing the VHH sequence. When non-pathogenic E. coli strains are provided with these adhesives, they colonize solid tumors expressing a target antigen more successfully than wild-type strains. Some compounds in the tumor microenvironment can act as natural chemoattractants for bacteria, such as vascular endothelial growth factor (VEGF), which is overproduced by cancer cells and attracts non-pathogenic E. coli strains [84]. Targeting the neoplasm area can also be enhanced by designing nutrient-deficient bacterial strains that can extract the necessary substances (for example, amino acids) from the tumor microenvironment. The S. typhimurium A1-R strain, auxotrophic for leucine and arginine, has demonstrated impressive therapeutic efficacy in mouse models of various types of cancer, including prostate, breast, pancreatic, ovarian, sarcoma, and glioma, leading to a slowdown in tumor growth compared to control groups [85]. 2.3. Bacteria Invasion of Tumor CellsIn some cases, the most advantageous strategy would be direct penetration of the bacterial courier directly into the cancer cell, for the subsequent release of the therapeutic cargo. To enter the tumor cell, bacteria can be equipped with an "invasion module", which consists of the constitutively expressed Yersinia pseudotuberculosis invasin gene (inv). Invasin binds to β1-integrins, which are overproduced on the surface of target cells in different types of tumors, and triggers bacterial capture through phagocytosis [86] (Fig. 5). In addition to the inv gene, the plasmid can include the the hlyA gene from Listeria monocytogenes, which codes for cytolysin. Cytolysin helps to destroy the phagosomal membrane and releases the therapeutic agent into the cytoplasm. This strategy was used to deliver transgenic interfering RNAs loaded into a non-pathogenic E. coli strain against the oncogenic HPV16-E7 protein in squamous cell carcinoma cells of the oral cavity [87].
2.4. Get it at All Costs: the Stealthy and Secure Courier Microbe
As mentioned above, bacteria can migrate in solid tumors until they reaches necrotic areas due to their hypoxic tropism and chemotaxis. At that, the immunosuppressive environment in these areas creates favorable conditions for bacterial reproduction. The rapid growth of bacterial population inside the tumor activates the host immune system and attracts immune cells. In addition, in order to reach tumors located in parts of the body that are usually considered sterile (such as blood, lungs, bladder, liver, bones etc.), bacteria need to overcome the immune defenses of the host in blood vessels and normal tissues. Bacteria, even if they are unable to reproduce, are foreign and toxic to the host; this raises reasonable questions about the effectiveness of using them as a therapeutic delivery vehicle. One of the ways to overcome the immunogenicity and toxicity of live couriers is the genetic knockout of the immunogenic elements of bacterial membranes; however, this strategy can lead to a significant weakening of the strain and a decrease in the effectiveness of colonization [88-91]. An alternative option is to mask microbial surfaces with non–immunogenic molecules, such as alginate-chitosan coatings mentioned in the previous section [54, 92], polydopamine [93], biocompatible lipids [94], or polymer nanoparticles [95]. An interesting approach to neutralizing the interaction between a bacteriobot and a host organism based on the programmed encapsulation of a bacterial cell, was proposed in by Harimoto et al. [96]. The authors modified the E. coli Nissle 1917 strain to control the expression of polysaccharide capsule genes using the external inducer, IPTG (isopropyl-β-D-1-thiogalactopyranoside). The regulated and dynamic synthesis of the polysaccharide capsule provided temporary protection for the bacteria as they moved towards the tumor, allowing them to evade the host's immune system. However, once their mission was completed, the loss of the capsule resulted in the effective elimination of the microorganisms. 3. HEAVILY ARMED: THE THERAPEUTIC AND DIAGNOSTIC POTENTIAL OF THE BACTERIAL ROBOT Among others, the most important characteristic of a bacterial microrobot is so-called "cargo" that it can deliver. So-called, because the bacterium itself can play the role of not only a carrier of various therapeutic molecules, but also a "living projectile" carrying immunogenic components on its surface, or a mobile factory for the production of drugs or prodrugs directly in the tumor area. As has been mentioned above, bacteria can stimulate a strong immune response in tumor microenvironment, attracting and activating immune cells and so providing an anti-tumor effect. In addition to naturally occurring immunogenic factors, such as peptidoglycan, lipopolysaccharides, and lipoteichoic acids, genetically modified bacteria can also produce additional immunomodulatory components aimed at stimulating the immunosuppressive environment of the tumor [47, 97-99]. For example, a therapeutic strain based on the non-pathogenic E. coli Nissle 1917 strain, developed by Leventhal et al., expresses cyclic diadenine monophosphate (cyclic diAMP, CDA) to activate one of the immune signaling pathways, STING [100]. The natural metabolism of bacteria can also be directed against the tumor, since multiplying bacteria compete with cancer cells for nutrients, can change the pH of the tumor microenvironment, or produce specific metabolites that inhibit the tumor cells growth, such as toxins, ROS, etc. [88, 101-102]. These properties can be further enhanced through genetic modification. A bacterium that has reached a tumor can serve as a source of continuous export of microRNA (miRNA) to "turn off" genes associated with the proliferation of tumor cells in RNA interference therapy. An example of this approach is an attenuated strain of S. typhimurium carrying a plasmid with a short hairpin RNA to suppress the expression of programmed cell death ligand 1 (PD-L1), a protein involved in mechanisms of evasion from immune control by tumor cells [103]. In combination with radiation therapy, this method has shown significant inhibition of tumor cell proliferation. As natural protein microfactories, bacteria can produce and secrete in the tumor microenvironment antibodies, enzymes, inflammatory factors or proteins that block or modulate metabolism, or inducers of apoptosis. In another well-known approach, a bacterium can encode an enzyme that catalyzes the conversion of a prodrug into a pharmacologically active antitumor compound. The advantage of this approach is that the active drug can be highly toxic, while its introduction into the body in the form of a prodrug reduces the risk of side effects. Finally, the bacterium can serve as a courier delivering a container with antitumor agent [5, 104-106]. It should be noted that researchers are showing amazing ingenuity in their attempts to create a universal and highly effective weapon from bacteria. The latest trend is to design bacterial robots with multiple functions, endowing them with a combination of different therapeutic and diagnostic tools. For example, one bacteriobot could combine the capabilities of immunotherapy with chemotherapy, or with the benefits of approaches such as radiotherapy (RT), photothermal (PTT) or photodynamic (PDT) therapy [91, 107-109]. For example, a complex and exciting sample of a multifunctional (multimodal) bacterial biohybrid combining the possibilities of chemotherapy with metabolic bacterial therapy was presented in [110], where the non-pathogenic strain of S. oneidensis MR-1 was equipped with "containers" of the MIL-101-MOF metal-organic framework loaded with doxorubicin. The Shewanella belongs to the electrogenic, bacteria, it has the ability to participate in electron transfer processes, and the strain MR-1 selectively metabolizes lactic acid as an energy source. The utilization of excess lactate in the tumor area makes it possible to improve the results of chemotherapy, while conversion Fe3+, a key element of the organometallic framework, to Fe2+ leads to the degradation of MIL-101-MOF and the release of doxorubicin. Fe2+ cations can further interact with hydrogen peroxide in the tumor zone to form hydroxyl radicals, which also have a damaging effect on cells, and the resulting Fe3+ ions further participate in lactate catabolism. More examples of multimodal bacteriobots can be found in recent excellent reviews [107, 111-115] and in Table 1. 4. THE SAFETY One of the most significant aspects of the idea of creating biological robots for the controlled delivery of therapeutic agents is biosafety. This includes not only safety for humans but also other aspects such as non-toxicity to the environment, the ability to degrade and be recycled after the mission, and biocompatibility. Biocompatibility aims at minimizing the effects of a long-term presence in the host body and reduce the impact on normal tissues. The main structural element of a bacteriobot, a bacterium, is usually of the greatest concern. Bacteria have the potential to multiply uncontrollably within a macroorganism and trigger an infectious process, producing substances that are toxic to tissues. Even if the bacterium is no longer viable or unable to reproduce, its DNA can still pose a threat because it can transfer genetic material to other strains that inhabit the human microbiome. One of the strategies for "biological containment" is the creation of auxotrophic strains. This involves removing a gene from the bacterial genome that encodes a metabolite essential for its survival. As a result, the viability of the bacterium is limited to regions where the necessary component can be obtained from outside the cell [116]. The SYNB1891 strain based on E coli Nissle 1917, auxotrophic in thymidine and diaminopimelic acid as a result of removal of the thyA and dapA genes, respectively, was constructed as a prototype of an antitumor agent [100]. Diaminopimelic acid is a component of the cell wall in gram-negative bacteria and is absent in eukaryotic cells, so a strain auxotrophic for this compound cannot survive in mammalian tissues for long time. Thymidine deficiency, commonly observed in extracellular environments, can also serve as an effective means of controlling bacterial behavior. The susceptibility of the bacterial robot to antibiotics seems to be a crucial requirement for biosafety management. In order to prevent the development of resistance while bacteria are staying in the human body, genes or gene cassettes for antimicrobial resistance can be pre-deleted or blocked, as done in [100]. The list of methods for controlling bacterial populations in the host body also includes the use of suicide genes triggered by an external signal [104], or the "deadman kill switch" proposed by Chan et al. [117]. This is a genetically engineered protective system that links environmental signals to suppressing or stimulating the production of toxins that are fatal to bacterial cells. Another approach to ensure the safety of microbial-based therapies is the development of bacteria with a reduced genome. This method involves removing non-essential coding sequences from the bacterial genome. The minimal genome only contains the genes that are necessary for the vital activity of the microorganism, effectively eliminating the possibility of bacterial adaptation to changing environmental conditions [118]. 5. BACTERIAL ROBOT... WITHOUT BACTERIA: THE PROSPECTS To create a bacterial robot, it is not necessary to use the whole bacterium. Instead, its parts or structural components can be used; this allows to increase safety in therapy while still benefiting from the use of a biological carrier. The potential of various bacterial components, such as bacterial mini-cells, membrane vesicles, magnetosomes, "bacterial ghosts" or spores, has been studied extensively. Bacterial mini-cells are nanoscale (100-400 nm in diameter), non-living, nuclear-free, non-dividing cells that are formed as a result of mutations in the minCDE genes [119]. These mutations lead to disruptions in the septa formation; as a result, there is more than one septa in the cell, or it located not in the center, but closer to the poles. Mini-cells contain all the components of the parent cell, except for the chromosomal DNA. Despite the fact that the mini-cells cannot multiply, they retain their residual metabolic activity. The main advantage of mini-cells is considered to be their ability to effectively encapsulate a drug inside themselves: for example, one mini-cell can contain up to a million molecules of doxorubicin, which is about a hundred times more than the liposome loading volume [119]. A bacterial ghost is the empty shell of a bacterial cell, primarily from gram-negative species. It lacks cytoplasmic components, but it retains a membrane with a complete set of antigenic protein structures [120]. These bacterial similarities can result from the expression of the E gene from the bacteriophage øX174 within the target cell. This expression produces a lysine protein that forms a transmembrane channel with a diameter up to 200 nm, causing the leakage of cytoplasmic content due to the osmotic pressure difference between the inside and outside of the cell membrane. Such structures - mini-cells and bacterial ghosts - are similar in that they are unable to multiply and actively metabolize, but they retain, in whole or in part, the cell morphology and antigenic surface elements - membrane proteins, adhesins, lipopolysaccharides, and peptidoglycan. Moreover, these structures can be further modified to target cancer cells more effectively, and the empty space within the cell membranes can be used to carry useful therapeutic cargo. Bacterial outer membrane vesicles (OMVs), nanoscale (20-300 nm) spherical bilayer lipid structures formed from fragments of the outer membrane and cell wall of Gram-negative bacteria, have been actively studied. In contrast to live bacteria, OMVs lack virulence but have high immunogenicity, stimulating natural immunity. Additionally, OMVs can enter host cells by fusion with cell membranes or endocytosis, increasing the efficiency of drug delivery [121]. The use of bacterial spores for delivering therapeutic agents to tumor areas is also promising. Bacterial spores, which are essentially a guarantee of species survival in adverse environments, are highly resistant to mechanical, chemical, and temperature changes, making them a powerful tool for delivering medicines within the human body. In hypoxic tumor foci, the spores of anaerobic bacteria can enter a vegetative state to synthesize and secrete therapeutic proteins [122]. In previous chapters, magnetotactic bacteria, which are capable of synthesizing unique organelles called magnetosomes inside their cells, were mentioned. Magnetosomes are crystals of iron oxide or sulfide surrounded by a phospholipid membrane. These organelles can be extracted from bacterial cells and used for targeted drug delivery in cancer therapy under the influence of a magnetic field [123]. Table 3 provides some examples of the potential of different biological carriers of bacterial origin.
CONCLUSIONS Bacteria are permanent companions of the human body throughout life and even after death. These tiny living beings, invisible to the naked eye, have evolved to coexist with humans and have developed various tools for interaction with the body. Therefore, the idea of creating bacteria-based robots that can freely move inside the body seems promising. However, despite numerous studies and some progress, the use of this therapy in clinical practice is not yet routine. The main challenge is to make bacteria's behavior completely predictable and ensure reliable control of the safety and efficacy of this potential therapeutic tool. Nevertheless, recent years have been characterized by significant progress towards the transformation of a natural bacterium into an ideal medical microrobot with desired properties. Some genetically modified bacterial strains are currently undergoing the last stages of clinical trials that precede the registration of drugs for cancer therapy [8, 31, 47, 91, 97, 107]. The main advantage of bacteriobots over traditional methods of cancer treatment is their ability to combine different methods of targeting tumor tissue within one miniature device. This allows for a more comprehensive approach to cancer treatment, providing a more effective and targeted solution. The development and manufacturing of bacterial microrobots is a complex process that requires integration of various technologies from different research fields, such as microbiology, genetic engineering, nanotechnology, biotechnology, synthetic biology, and microfluidics. Additionally, artificial intelligence can be useful to predict the properties of these robots, and clinical medicine is necessary to evaluate their performance. In some works [86, 104], bacteria and bacteria-based biohybrids are proposed to be viewed as a combination of unique modules that each perform specific functions. At that, the characteristics of each module can be modified as needed for different tasks. In conclusion, it should be noted that while there is still much work to be done before bacterial biohybrid drugs can be routinely used in clinical practice, with a sufficient number of laboratory prototypes. There are many questions that remain unanswered, such as the pharmacokinetics and pharmacodynamics of these drugs in the human body, which are currently not well understood. However, this path has a definite potential. COMPLIANCE WITH ETHICAL STANDARDS Not applicable. ACKNOWLEDGEMENTS The author is grateful to O.L. Kurbatova for her invaluable help with the design of the drawings for the article, and to L.K. Kurbatov for fruitful discussions and useful comments. FUNDING The authors declare that they received no external funding. CONFLICT OF INTEREST The authors declare that they have no conflicts of interest. REFERENCES
|